As with any medicine, the MHRA will keep the safety and effectiveness of polihexanide under close review.
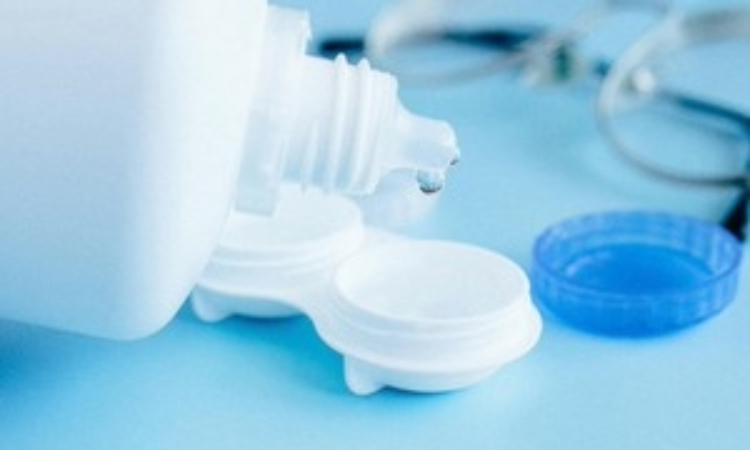
The Medicines and Healthcare products Regulatory Agency (MHRA) has approved polihexanide (Akantior) to treat acanthamoeba keratitis.
Acanthamoeba keratitis is an infection of the cornea, the clear ‘window’ at the front of the eye, that can be very painful.
This medicine has been approved through the International Recognition Procedure (IRP). The IRP allows the MHRA to take into account the expertise and decision-making of trusted regulatory partners for the benefit of UK patients.
Polihexanide is administered as an eye drop solution, directly into the eye.
The MHRA conducts a targeted assessment of IRP applications and retains the authority to reject applications if the evidence provided is not considered sufficiently robust.
A full list of side effects can be found in the Patient Information Leaflet (PIL) or the Summary of Product Characteristics (SmPC), available on the MHRA website within 7 days of approval.
As with any medicine, the MHRA will keep the safety and effectiveness of polihexanide under close review.
Anyone who suspects they are having a side effect from this medicine is encouraged to talk to their doctor, pharmacist or nurse and report it directly to the MHRA Yellow Card scheme, either through the website (https://yellowcard.mhra.gov.uk/) or by searching the Google Play or Apple App stores for MHRA Yellow Card.
Notes
- The approval was granted on 15 June 2025 to SIFI S.P.A.
- This product was submitted and approved via International Recognition Procedure.
- More information can be found in the Summary of Product Characteristics and Patient Information leaflets which will be published on the MHRA Products website within 7 days of approval.
- The Medicines and Healthcare products Regulatory Agency (MHRA) is responsible for regulating all medicines and medical devices in the UK by ensuring they work and are acceptably safe. All our work is underpinned by robust and fact-based judgements to ensure that the benefits justify any risks.
- The MHRA is an executive agency of the Department of Health and Social Care.
source: GOV

 ENFIELD
ENFIELD HACKNEY
HACKNEY HARINGEY
HARINGEY ISLINGTON
ISLINGTON