The MHRA has advised pharmacy and healthcare professionals to stop supplying the impacted batch and return all remaining stock to their suppliers.
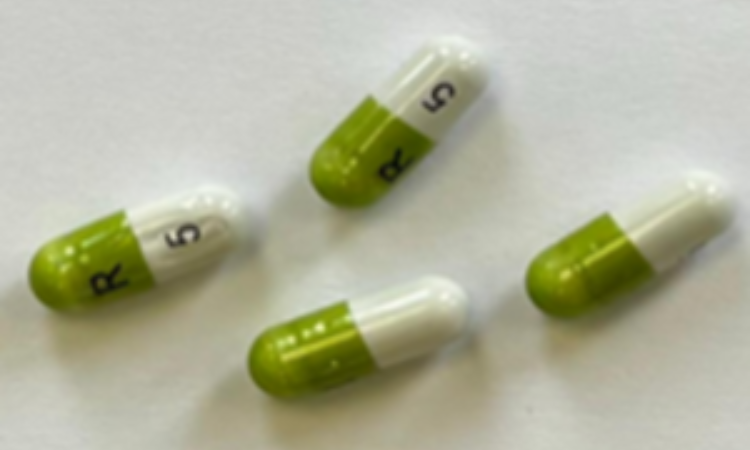
Crescent Pharma Limited is recalling one batch of Ramipril 10mg capsules as a precautionary measure due to a potential manufacturing error which may mean some cartons contain blister strips of a lower dose, specifically Ramipril 5mg.
This follows a complaint from a patient where it was identified that, inside a sealed carton of Ramipril 10mg capsules, one blister pack of Ramipril 5mg capsules was found. Both product batches were manufactured at the same manufacturing site, and the error appears to have occurred during secondary packaging of the cartons.
The risk to patients of taking the lower dose of this medicine for a limited time is very low.
Dr Alison Cave, MHRA Chief Safety Officer, said:
“If you take Ramipril 10mg, check the packaging for batch number GR174091. The batch number and expiry date information can be found on the outer carton. If you have received this batch, check that the medication name on the carton matches the blister strips inside.
“If the 10 mg carton of Ramipril contains blister strips that are labelled as Ramipril 5mg capsules, contact your dispensing pharmacy. If the carton contains blister strips that are correctly labelled as Ramipril 10mg capsules, you do not need to take further action.”
If you have an impacted pack or previously received this batch and you believe you have taken any Ramipril 5mg capsules that were included in error and are currently experiencing any adverse effects, please seek medical advice. Please take the leaflet that came with your medicine and any remaining tablets with you to your pharmacy or GP practice. Any suspected adverse reactions should also be reported via the MHRA Yellow Card scheme.
If you’ve already taken Ramipril 5mg, please be reassured that there is a very low risk to your health. Both strengths of the medication are used to treat high blood pressure, heart failure, and kidney disease. Any possible impact of a lower dose of Ramipril is expected to be gradual rather than immediate or life threatening.
The MHRA has advised pharmacy and healthcare professionals to stop supplying the impacted batch and return all remaining stock to their suppliers.
Notes
- Please see MHRA’s Class 2 recall for further information and images of the affected product.
- The MHRA is responsible for regulating all medicines and medical devices in the UK by ensuring they work and are acceptably safe. All our work is underpinned by robust and fact-based judgements to ensure that the benefits justify any risks.
- The MHRA is an executive agency of the Department of Health and Social Care.
source: GOV

 ENFIELD
ENFIELD HACKNEY
HACKNEY HARINGEY
HARINGEY ISLINGTON
ISLINGTON